Cell-based therapies are rapidly advancing, driving demand for scalable, standardized manufacturing. ATLATL’s platform leverages CliniMACS® closed-system technology to process both autologous and allogeneic therapies with greater consistency and efficiency. Supporting both scale-out and scale-up, and adaptable to centralized or point-of-care models, the platform accelerates the path from development to commercialization.
CliniMACS® Plus Applications
- T cell isolation: CD4/CD8 positive selection
- T cell: TCR α/β depletion
- NK cells: CD3 depletion / CD56 enrichment
- Treg isolation: CD25 enrichment
- B cells: CD19 depletion or enrichment
- HSC isolation: CD34 enrichment
The CliniMACS Prodigy® platform
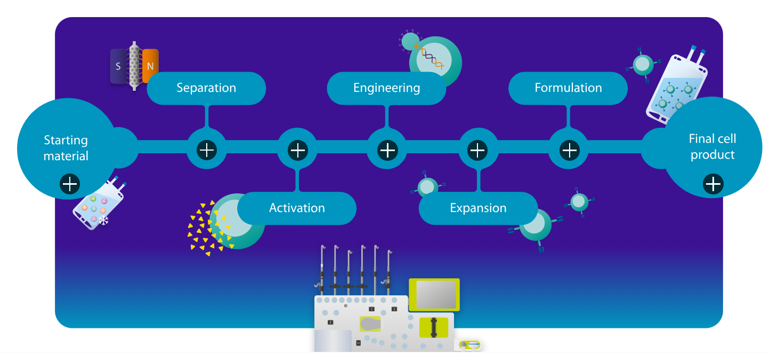
The CliniMACS Prodigy® platform integrates all key steps of T cell manufacturing into an automated, closed system, with workflows tailored to different therapeutic needs:
- T Cell Transduction (TCT): The standard approach for CAR-T manufacturing, enabling efficient viral transduction and robust gene delivery.
- T Cell Transduction with Automated Formulation (TCTf): Extends the TCT workflow with integrated formulation and fill capabilities for precise final product preparation.
- T Cell Transduction – Large Scale (TCT-LS): Scales the TCT workflow to larger culture volumes, supporting higher-dose and large-scale manufacturing.
Designed for GMP-compliant production, these workflows enable automated, standardized manufacturing of gene-engineered T cells. By minimizing manual handling and open steps, the platform ensures consistent, reproducible cell production suitable for clinical applications.
